Change Page: < 12345 > | Showing page 3 of 5, messages 41 to 60 of 83 - powered by ASPPlayground.NET Forum Trial Version
Author
|
Message

|
 Re:Breeding Journal, Species: Amphiprion ocellaris "Black / Darwin"
Saturday, February 27, 2010 9:40 PM
Re:Breeding Journal, Species: Amphiprion ocellaris "Black / Darwin"
Saturday, February 27, 2010 9:40 PM
( permalink)
Sanjay, that IS interesting. I know that if I put back a tile that has died in artificial incubation, the parents will remove the dead eggs and eat them. I've never tried giving back a partially hatched tile. Someday, I should try that. It also speaks to the relative resilience of clownfish eggs, that is to say that the water chemistry in the hatching tank could diverge from the broodstock tank, yet the eggs can withstand the move, sans acclimation, without much consequence. HMM...very interesting. On a Black Ocellaris note. This morning, another gallon of sieved rotifers went in. Mutiple times today, I offered light dustings of Otohime A for food. This evening, 1 more gallon of clean new water was added along with 20 drops of RotiGreen, 5 drops of Chloram-X, and 2 more gallon's worth of seived (53 micron) rotifers. Bottom line, I'm very happy that I now have 3 productive rotifer cultures going...the clown babies are really plowing through them now! Settlement sometime in the coming week.. 
|
|
|
 Re:Breeding Journal, Species: Amphiprion ocellaris "Black / Darwin"
Monday, March 1, 2010 11:35 AM
Re:Breeding Journal, Species: Amphiprion ocellaris "Black / Darwin"
Monday, March 1, 2010 11:35 AM
( permalink)
So between yesterday and now, I've added water, drained down to 5 gallons, added 20 drops of RotiGreen yesterday and another 10 this morning, added 10 drops of Chloram-X, Harvested 2 gallons of Rotifers last night and another 2 gallons this morning, and have periodically added small feedings of Otohime A. No noticed deaths now for at least 24 if not 48 hours. Things are looking solid. I feel like I should have headstripes soon...
(they are 7+ and 6+ days post hatch, FWIW).
|
|
|
 Re:Breeding Journal, Species: Amphiprion ocellaris "Black / Darwin"
Monday, March 1, 2010 11:25 PM
Re:Breeding Journal, Species: Amphiprion ocellaris "Black / Darwin"
Monday, March 1, 2010 11:25 PM
( permalink)
More fun...we're at basically 7 to 8 days post hatch, and I've noticed my first "waddle" swimming this evening. Meta is coming soon!
Harvested a "whopping" 3 gallons of rotifers this evening as well as adding in 10 drops of RotiGreen (tank is running at about 6 gallons right now). Plan is to drip in maybe another 10 drops of RotiGreen, some Chloram-X, and 2 gallons of clean new saltwater. We'll see if that all gets done yet tonight!
FWIW,
Matt
|
|
|
 Re:Breeding Journal, Species: Amphiprion ocellaris "Black / Darwin"
Tuesday, March 2, 2010 1:08 AM
Re:Breeding Journal, Species: Amphiprion ocellaris "Black / Darwin"
Tuesday, March 2, 2010 1:08 AM
( permalink)
2:05 AM, March 2, 2010 - WE HAVE HEADSTRIPES!!!
I just looked in to make sure the last of the new water dripped in, and I noticed at least 3 larvae hanging at the sides with headstripes, only visible when looking in from above. I'm going to refrain from taking pictures at this time as I don't want to shock or stress the babies in this state, so you'll have to trust me (unless I get some good pictures without a flash...)
|
|
|
 Re:Breeding Journal, Species: Amphiprion ocellaris "Black / Darwin"
Tuesday, March 2, 2010 1:27 AM
Re:Breeding Journal, Species: Amphiprion ocellaris "Black / Darwin"
Tuesday, March 2, 2010 1:27 AM
( permalink)
Right on time, cool!
|
|
|
 Re:Breeding Journal, Species: Amphiprion ocellaris "Black / Darwin"
Tuesday, March 2, 2010 9:57 PM
Re:Breeding Journal, Species: Amphiprion ocellaris "Black / Darwin"
Tuesday, March 2, 2010 9:57 PM
( permalink)
First, settlement pictures, shot early morning 3-2-2010.   Second, on the evening of 3-2-2010, another massive spawn on the Black O's...
|
|
|
 Re:Breeding Journal, Species: Amphiprion ocellaris "Black / Darwin"
Wednesday, March 3, 2010 7:46 AM
Re:Breeding Journal, Species: Amphiprion ocellaris "Black / Darwin"
Wednesday, March 3, 2010 7:46 AM
( permalink)
Last night, trickled in another gallon of clean new water with a little Reef Plus and Reef Iodide. This morning, as is customary now I offered up Otohime A. I also enriched 2 gallon's worth of rotifers with Super Selcon for 30 minutes before rinsing and feeding it in....the fish are not weaning as easy as I'd like, so I'm continuing to offer up rotifers while trying to get them straight onto the Otohime. No hatching brine shrimp for me!
There is somewhere between 30 and 50% headstripes at this point. I've seen one baby that is clearly deformed and will need to be culled. I've also seen one or two babies that already have the start of the SECOND stripe, just a tiny white dot at the back at this point.
FWIW,
Matt
|
|
|
 Re:Breeding Journal, Species: Amphiprion ocellaris "Black / Darwin"
Wednesday, March 3, 2010 8:01 AM
Re:Breeding Journal, Species: Amphiprion ocellaris "Black / Darwin"
Wednesday, March 3, 2010 8:01 AM
( permalink)
Oh, and BTW, as far as I'm concerned, we'll probably be updating that Bio of mine to say "successfully reared 6 species" instead of "5" 
|
|
|
 Re:Breeding Journal, Species: Amphiprion ocellaris "Black / Darwin"
Thursday, March 4, 2010 9:39 AM
Re:Breeding Journal, Species: Amphiprion ocellaris "Black / Darwin"
Thursday, March 4, 2010 9:39 AM
( permalink)
3-4-10 - 10:35 AM
80% have gone through Meta at this point in time. Yesterday, drained the tank back down to half full (5 gallons), scraped the bottom and sucked out lots of "gunk". Trickled in 2 gallons of new saltwater at around 1.023. This morning, fed Otohime A, fed more Otohime A, started another trickle of clean water in (aka we were at 7 gallons this AM, and I'll probably bring it back up to 9 by midday). Fed in more rotifers, just for the stragglers. Have a few fish now showing the starts of 2nd stripe development, starting along the back of the fish.
Interesting to note, that since I've raised these fish so far with the lights on 24/7, they are very STRONGLY phototrophic. I turned the lights off for a moment last night, and the entire mass of babies bumrushed the one side panel of the tank that isn't painted black. I think I'll have to ease the fish gently onto a day night cycle.
FWIW,
Matt
|
|
|
 Re:Breeding Journal, Species: Amphiprion ocellaris "Black / Darwin"
Thursday, March 4, 2010 10:03 PM
Re:Breeding Journal, Species: Amphiprion ocellaris "Black / Darwin"
Thursday, March 4, 2010 10:03 PM
( permalink)
Giving them their first "night"...room lights staying on, but I blocked off the unpainted end so that light is only coming in from above. They kinda freaked.
Also, once again brought them down to 5 gallons of water, and trickled in one new clean gallon. Will trickle more in tomorrow. Still around 80-90% settlement.
|
|
|
 Re:Breeding Journal, Species: Amphiprion ocellaris "Black / Darwin"
Friday, March 5, 2010 12:39 AM
Re:Breeding Journal, Species: Amphiprion ocellaris "Black / Darwin"
Friday, March 5, 2010 12:39 AM
( permalink)
I'm curious, why did you choose to light them 24/7? Also, why did you not cover the non painted end of the tank until you changed the lighting cycle?
Is this a "Darwin" specific experiment?
The reason I ask is that from what I've read and my own experience with various Amphiprion species, 24/7 lighting has no positive/beneficial effect.
In contrast, 24/7 lighting has worked well with my P. fridmani...
|
|
|
 Re:Breeding Journal, Species: Amphiprion ocellaris "Black / Darwin"
Friday, March 5, 2010 1:49 PM
Re:Breeding Journal, Species: Amphiprion ocellaris "Black / Darwin"
Friday, March 5, 2010 1:49 PM
( permalink)
 Originally Posted by Fishtal I'm curious, why did you choose to light them 24/7 Faster growth.
 Originally Posted by Fishtal Also, why did you not cover the non painted end of the tank until you changed the lighting cycle? At night, with the 24/7 lighting, the only lighting coming in was from above. But once I turned out the main light, and left the room lights on, the babies instantly went for the unpainted side where light was coming in. Thus, blocking it kept the strongest light coming in from above.
 Originally Posted by Fishtal Is this a "Darwin" specific experiment? Not really...just figured I might as well do it since it had a positive impact on some other species.
 Originally Posted by Fishtal The reason I ask is that from what I've read and my own experience with various Amphiprion species, 24/7 lighting has no positive/beneficial effect. In contrast, 24/7 lighting has worked well with my P. fridmani...
Again, other species efforts led me to try it here just to see. As of today, maybe 95% through meta. Lost one smaller one overnight. Headstripes all over the place, and 2nd stripes forming on maybe 10-20 individuals now... Matt
|
|
|
 Re:Breeding Journal, Species: Amphiprion ocellaris "Black / Darwin"
Monday, March 8, 2010 11:35 PM
Re:Breeding Journal, Species: Amphiprion ocellaris "Black / Darwin"
Monday, March 8, 2010 11:35 PM
( permalink)
First, pictures shot the morning of 3-7-10 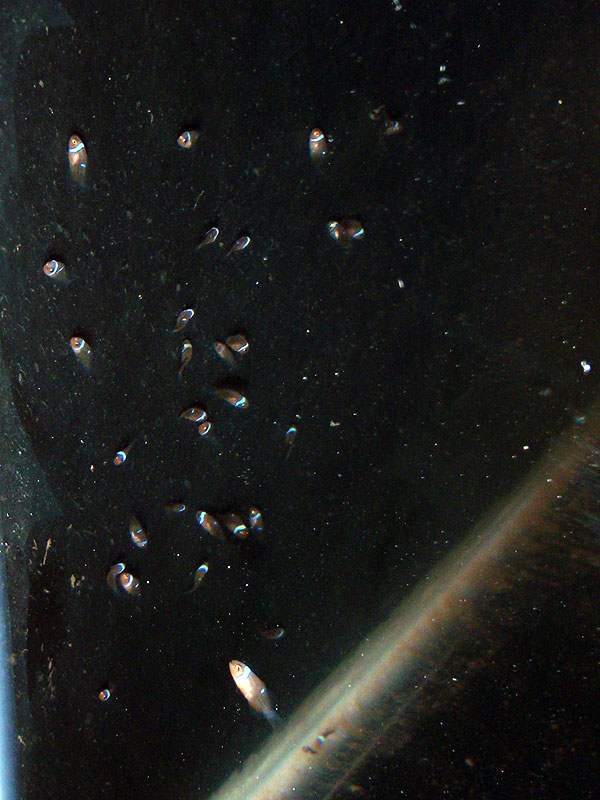  This evening, I moved the babies into a different 10 gallon tank. This tank has an established internal filter, which I threw in a breeder net so that it doesn't suck up the babies  Made sure that the back side of the net is kept away from the back of the tank with a rigid airline tubing so that babies don't get stuck back there. During the move, I took a headcount. Officially, 181 baby Black O's. Shot these just after midnight, wee hours of the morning on 3-9-10, the move and right after:    
|
|
|
 Re:Breeding Journal, Species: Amphiprion ocellaris "Black / Darwin"
Saturday, March 20, 2010 11:15 PM
Re:Breeding Journal, Species: Amphiprion ocellaris "Black / Darwin"
Saturday, March 20, 2010 11:15 PM
( permalink)
Grats on the Black O's Matt. I am curious as to why you did not feed bbs and tried to go straight from rotifers to Otohime A? I have had good success with weaning to Otohime but always from bbs. Also do you think the Cloram-x has the potential to have any negative effects on the juvenilles?
Dave
|
|
|
 Re:Breeding Journal, Species: Amphiprion ocellaris "Black / Darwin"
Wednesday, March 24, 2010 12:38 PM
Re:Breeding Journal, Species: Amphiprion ocellaris "Black / Darwin"
Wednesday, March 24, 2010 12:38 PM
( permalink)
Dave, I don't have hard answers to your questions. Skipping BBS is simply something you CAN DO with Otohime A, and in theory, Otohime is more nutritionally appropriate than BBS, so better to just go straight to it. I've been told by the fine folks @ Reed that Chloram-X has no ill effects on fish larvae.
As of 3-24-10, I'm back in Duluth and there are many babies getting tailstripes. It looks like this batch is OVERWHELMINGLY Misbars...very sad turn of events. There are many babies I can clearly see that I'll need to cull, and frankly part of me is wondering if ANY of the batch will really be salable fish. Certainly a big disappointment, and clearly I need to improve my larval rearing!
|
|
|
 Re:Breeding Journal, Species: Amphiprion ocellaris "Black / Darwin"
Wednesday, March 24, 2010 8:31 PM
Re:Breeding Journal, Species: Amphiprion ocellaris "Black / Darwin"
Wednesday, March 24, 2010 8:31 PM
( permalink)
 Originally Posted by mPedersen Interesting to note, that since I've raised these fish so far with the lights on 24/7, they are very STRONGLY phototrophic. I turned the lights off for a moment last night, and the entire mass of babies bumrushed the one side panel of the tank that isn't painted black. I think I'll have to ease the fish gently onto a day night cycle. Very cool Matt, congrats on the Darwins, are they still afraid of the dark?
|
|
|
 Re:Breeding Journal, Species: Amphiprion ocellaris "Black / Darwin"
Wednesday, March 24, 2010 9:02 PM
Re:Breeding Journal, Species: Amphiprion ocellaris "Black / Darwin"
Wednesday, March 24, 2010 9:02 PM
( permalink)
 Originally Posted by mPedersen
Dave, I don't have hard answers to your questions. Skipping BBS is simply something you CAN DO with Otohime A, and in theory, Otohime is more nutritionally appropriate than BBS, so better to just go straight to it. I've been told by the fine folks @ Reed that Chloram-X has no ill effects on fish larvae.
As of 3-24-10, I'm back in Duluth and there are many babies getting tailstripes. It looks like this batch is OVERWHELMINGLY Misbars...very sad turn of events. There are many babies I can clearly see that I'll need to cull, and frankly part of me is wondering if ANY of the batch will really be salable fish. Certainly a big disappointment, and clearly I need to improve my larval rearing!
Paragraph 1: I almost always skip BBS and go right to Otohime A with no ill effects. You mentioned Chloram-X a few times while you were here and I just recently heard a few people mention problems with using Amquel... can you point out any discussion, or thoughts, on that? Paragraph 2: My first batch of Darwins was about 70% misbars, the next batch was less than 20%. Not sure what happened since I did the same thing for both batches... I attributed it to being the first batch. I was told by all of my stores that misbarred Darwins had no bearing on sales, unless there was something really cool, lol. I don't want to get into a discussion about culling here, maybe we should start a thread on that?
|
|
|
 Re:Breeding Journal, Species: Amphiprion ocellaris "Black / Darwin"
Wednesday, March 24, 2010 10:35 PM
Re:Breeding Journal, Species: Amphiprion ocellaris "Black / Darwin"
Wednesday, March 24, 2010 10:35 PM
( permalink)
Yeah, I know many folks LIKE or perhaps "don't care" about misbars, but I do. I DISLIKE them. I think though, that I had problems beyond misbarring, i.e. there are a fair number off outright culls (i.e. stubbies, kinked backs, clearly flared gills) so I think that even with the daily water changes the water quality didn't work out.
Regarding Amquel Issues, the first thing I would ask is are they "chemically the same"? I.e. there may be chemicals in one that aren't the other, and that could be the problem. Again, it could just as easily be that there ARE issues with using Chloram-X, but again, what I was told by Reed is that there aren't.
And no, the Darwins are no longer afraid of the dark...they're on a regualr day night schedule now. STILL strongly phototrophic though!
|
|
|
 Re:Breeding Journal, Species: Amphiprion ocellaris "Black / Darwin"
Thursday, March 25, 2010 10:35 PM
Re:Breeding Journal, Species: Amphiprion ocellaris "Black / Darwin"
Thursday, March 25, 2010 10:35 PM
( permalink)
Matt, Thanks for the feedback. I recently hatched a batch of A. Ocellaris and they are on day 5 now. I have always introduced bbs to the larva on either day 5 or 6 and then introduced Oto A on day 13 or 14 with a 7 day overlap in foods. Typically the babies are off rotifers by day 14 and bbs by day 21 with them being completely weaned to dry foods. I have never put an ammonia neutralizer into the larval water but am experimenting with this batch. This batch is relatively small (approximately 100 fish) but I am really wanting to see if the cloram-x will make a difference in the markings on the fish. These babies will receive the exact same regimen in regards to foods and water changes as normal to see if the cloram-x will make a difference. I also keep the lighting on 24/7 on the larva thru meta and believe it does make a difference especially in the first few days of the larval cycle. I also believe that the larva start into meta sooner when the lighting is on 24/7. Thanks again for introducing me to Tal. Without the introduction I wouldn't have found this site  Dave
|
|
|
 Re:Breeding Journal, Species: Amphiprion ocellaris "Black / Darwin"
Friday, March 26, 2010 4:38 PM
Re:Breeding Journal, Species: Amphiprion ocellaris "Black / Darwin"
Friday, March 26, 2010 4:38 PM
( permalink)
Dave, it's absolutely my pleasure to have made the introduction. I'll be waiting with bated breath to hear the results of your ChloramX experiment. I'm still ticked that I've had such a high rate of misbars in this first batch. On the upside....while I was gone Renee reared 3 baby Black O's. They hatched on March 10th.
And now, March 26th, I have a new batch of eggs going down this evening  A chance to FIX my misbarring problem the first time around. I welcome suggestions on what I might have done wrong...
|
|
|
|