Change Page: < 1234567 > | Showing page 4 of 7, messages 61 to 80 of 130 - powered by ASPPlayground.NET Forum Trial Version
Author
|
Message

|
 Re:Breeding Journal, Species: Pseudochromis tapeinosoma
Tuesday, February 14, 2012 12:39 AM
Re:Breeding Journal, Species: Pseudochromis tapeinosoma
Tuesday, February 14, 2012 12:39 AM
( permalink)
It only takes one. 
|
|
|
 Re:Breeding Journal, Species: Pseudochromis tapeinosoma
Wednesday, February 15, 2012 6:48 PM
Re:Breeding Journal, Species: Pseudochromis tapeinosoma
Wednesday, February 15, 2012 6:48 PM
( permalink)
By the time I got home about 1:00 AM PST this morning, there were no larvae left that I could see. Bummers, but at least the fish sitter got at least one of them to 4 days. That's a start, I guess. The female is looking nice and plump, and will probably be ready to spawn again in just a day or two or three. This time, I'll be around to (I hope) do a more proper job of caring for them.
|
|
|
 Re:Breeding Journal, Species: Pseudochromis tapeinosoma
Friday, February 17, 2012 12:07 PM
Re:Breeding Journal, Species: Pseudochromis tapeinosoma
Friday, February 17, 2012 12:07 PM
( permalink)
Another spawn today, 12 days since the last spawn. Very much the same behavior as the last spawn. They went almost immediately into the spawning PVC cave, and stayed there together for about 1/2 hour, and then she almost immediately left through the hole in the divider back to her side. This time, I plan on using the BRT, copepods, and live ISO greenwater. I'm back from vacation, but have come down with the flu now, so I'll have time to do lots of reading (and re-reading) between now and then. Moe's dottyback book and forum posts on the reading list for the next few days!
|
|
|
 Re:Breeding Journal, Species: Pseudochromis tapeinosoma
Wednesday, February 22, 2012 12:46 AM
Re:Breeding Journal, Species: Pseudochromis tapeinosoma
Wednesday, February 22, 2012 12:46 AM
( permalink)
Did did a great job of caring for the eggs. Tonight is night #4 for this spawn. I've had a bunch of ASW that has been aging for almost 2 weeks while I was on vacation. I've been doing multiple small water changes over the last few days in the broodstock tank. I also prepared a bleached, dechlored, rinsed, and dried BRT with 5 gallons of the new, aged ASW. Made sure the temperature of both tanks matched. I set up a cut-down version of a brine shrimp hatcher/tumbler (brand new, thoroughly cleaned bottle, of course) in the BRT. Airstone under the heater in the BRT. Ready for lights out! Right after lights out, I removed the egg ball, or what was left of it, and placed it into the tumbler in the BRT. It was not so much an egg ball, as it was two or three loosely connected egg masses. "Nets" as Tal referred to it in one of his dottyback journals. The tumbling eggs were lit with a fluorescent strip light place right over the tumbler supported on some eggcrate that spanned the width of the BRT. Within minutes, I saw the first larvae. Within just about 5 to 10 minutes, I started seeing hundreds and hundreds of them! Again taking my cues from Tal's suggestions, I stopped the air in the tumbler from time to time, let the eggs settle out, and turkey basted the larvae out of the tumbler and into the BRT proper, replacing the water I removed from the tumbler with BRT water. I repeated this process for an hour or so, until it appeared that all of the eggs that were going to hatch did hatch, at which point I removed the tumbler and unhatched eggs. There were very, very few unhatched eggs. I took a dense culture of Apocyclops that I've been raising in a 2 gallon fishbowl (actually, these are the ones left from the last failed attempt by the fish sitter at raising them in the fishbowl kreisel -- the Apo left over from that experiment have done very, very well!) and sieved them out, and backwashed them into the BRT. I also took about 500 ml of dense live Isochrysis and slowly dripped it in, 1 drop per second. I'm leaving the lights on all night tonight, and will monitor the copepod density and phyto density closely. EDIT: I forgot to mention that I also added 2.5 ml of AmQuel Plus. I have some rotifers that are growing in one of my copepods buckets in the greenhouse outside, if I have to resort to using them, but I want to try to raise these larvae with copepods only, if possible. Here are some images: The eggs tumbling in the BRT:  A composite of stiched microscope images of one larvae:  The same larvae, taken with the DSLR and a lens on a bellows: 
<message edited by JimWelsh on Wednesday, February 22, 2012 1:41 AM>
|
|
|
 Re:Breeding Journal, Species: Pseudochromis tapeinosoma
Wednesday, February 22, 2012 12:53 AM
Re:Breeding Journal, Species: Pseudochromis tapeinosoma
Wednesday, February 22, 2012 12:53 AM
( permalink)
Jim,
Those photos are excellent.
What software do you use to stich the images together?
Im I right that the microscope images are back lit and the DSLR images are top lit?
|
|
|
 Re:Breeding Journal, Species: Pseudochromis tapeinosoma
Wednesday, February 22, 2012 1:07 AM
Re:Breeding Journal, Species: Pseudochromis tapeinosoma
Wednesday, February 22, 2012 1:07 AM
( permalink)
Darren,
Thanks. The microscope image has some nuisance algae in the way, but I was too busy to care. I used a program called "PhotoStitch" that came with my Canon Digital Rebel XT when I bought it about 6 years ago. It is very easy to use. PM me. Both are back lit.
|
|
|
 Re:Breeding Journal, Species: Pseudochromis tapeinosoma
Wednesday, February 22, 2012 1:07 AM
Re:Breeding Journal, Species: Pseudochromis tapeinosoma
Wednesday, February 22, 2012 1:07 AM
( permalink)
EDIT: Using accidental double post from last night for my next update. After 8 hours, I awoke to run downstairs and check on the babies. Looks like just about the same number swimming around as last night. Check temp -- 81.5 F -- a little warm, but OK. Check ammonia -- unmeasurable with API test kit -- yay! the AmQuel Plus and/or the Live Isochrysis are doing their jobs! Check SG -- no appreciable evaporation since last night. Check copepod density -- OH MY! These larvae can EAT. They have just about decimated the copepod population. I've scrambled and gotten together another gallon or so of dense Apocyclops to sieve out and feed them, but I'm probably going to be in some serious trouble here in a short period of time, if the population of the larvae stays high. I'd better start harvesting and enriching some rotifers from the buckets I have in the greenhouse!
<message edited by JimWelsh on Wednesday, February 22, 2012 9:42 AM>
|
|
|
 Re:Breeding Journal, Species: Pseudochromis tapeinosoma
Thursday, February 23, 2012 10:35 PM
Re:Breeding Journal, Species: Pseudochromis tapeinosoma
Thursday, February 23, 2012 10:35 PM
( permalink)
Right about 48 hours post hatch. There are fewer of them now, but certainly at LEAST 50% are still alive, and probably more like 75%. The ammonia is staying at 0, and the only thing I've added in the last two days is more sieved copepods, and tonight, just about an hour ago, one gallons's worth of sieved rotifers from a rotifer culture I'm nursing along that has maybe 15 rots / ml right now. The rots have been raised for the last day and a half on RGComplete, with no additional enrichment. The Iso in the BRT has diminished somewhat, which tells me that the copepods must be eating it. I'll add probably another 500 - 1000 ml of Iso tonight, since I'm sure the rotifers will be eating it up. The SG has gone up by about .002 in the last two days, and I'll be topping off with some RO/DI tonight, too, to bring it back down a bit. The temp is staying between 79 and 80.5 F. I sacrificed one to the microscope gods tonight, but am too lazy to do the DSLR/Bellows thing. This larva is not only larger, it is much more opaque than the newly hatched larva was. I'll need to use the DSLR/Bellows for the future shots, to be sure. Examining him with a powerful hand lens, I could clearly see a full gut. 
|
|
|
 Re:Breeding Journal, Species: Pseudochromis tapeinosoma
Friday, February 24, 2012 8:59 AM
Re:Breeding Journal, Species: Pseudochromis tapeinosoma
Friday, February 24, 2012 8:59 AM
( permalink)
After feeding more Iso and topping off with some RO, I turned the lights out on them and went to bed. Woke up this morning to almost all of them dead. There are some still alive, but I'd say I'm down to far less than 100 now.
I can think of only two things that might have caused this:
1) The previous night, I also turned the lights out, but that was after staying up very late, and working until almost 4:30 AM, so they only had about 2 hours of "dark" before sunrise and lights from other tanks coming on nearby would have given them ambient light. This time, they had a solid 7 hours of darkness. Perhaps that is the main cause. I have a heater with a red light in the BRT, and an airstone making a bubble curtain around that heater. Perhaps the light draws them to the bubbles, which then do bad things to the larvae?
2) I did use some older Iso as part of the feed last night. It was "past its prime", as in it was probably close to crashing, but had not crashed yet. Maybe that was the problem? Ammonia still tests at zero.
Due to the ammonia test results, I suspect #1 more than #2.
Disappointed, but not devastated yet. There's still some hope for getting at least one through in this batch. If the parents keep to their schedule, I should have another spawn in about 5 days or so now, even if this one goes further south.
EDIT: Note that the Iso and Top Off water were dripped, one drop per second, into the bubbles over the heater, and that the SG this morning is 1.025, down just 0.001 from last night. It wasn't shock from too much or too quick top off.
|
|
|
 Re:Breeding Journal, Species: Pseudochromis tapeinosoma
Friday, February 24, 2012 11:49 AM
Re:Breeding Journal, Species: Pseudochromis tapeinosoma
Friday, February 24, 2012 11:49 AM
( permalink)
I lit my P. fridmani 24/7 until settlement but I was only feeding them rotifers at the time.
|
|
|
 Re:Breeding Journal, Species: Pseudochromis tapeinosoma
Tuesday, February 28, 2012 12:52 AM
Re:Breeding Journal, Species: Pseudochromis tapeinosoma
Tuesday, February 28, 2012 12:52 AM
( permalink)
Why no rotifers Jim? Copepods would be fine as a supplement, and rotifers certainly should work for the mainstay. You saw how I did with Rotifers + Reed's pastes with Flavivertex....
|
|
|
 Re:Breeding Journal, Species: Pseudochromis tapeinosoma
Tuesday, February 28, 2012 1:00 AM
Re:Breeding Journal, Species: Pseudochromis tapeinosoma
Tuesday, February 28, 2012 1:00 AM
( permalink)
Matt, I did feed some rotifers on 2/23 (see post above). My rationale was that they were eating the copepods, and growing, and had full guts. My thought was that guts full of copepods > guts full of rotifers. Maybe I'll do both on the next batch.
At this point, I'm down to just a couple of larvae from this hatch. Since there are so few larvae, the copepods are doing VERY well, and I assume that they are adequate food for the few larvae left. My maintenance the last few days has been mostly to just add RO/DI to deal with evaporation. I do believe that the problem was somehow related to the long dark period.
The parents are due for another spawn in the next day or two. I have another BRT that I can ready for that hatch, and I have adequate copepods and rotifers for that batch (I think). I believe that I've learned from this batch, and hope to do better on the next one. I seem to have good parent broodstock, with a very diligent and competent male to tend the eggs for me until hatch time.
Stay tuned!
|
|
|
 Re:Breeding Journal, Species: Pseudochromis tapeinosoma
Wednesday, February 29, 2012 1:24 PM
Re:Breeding Journal, Species: Pseudochromis tapeinosoma
Wednesday, February 29, 2012 1:24 PM
( permalink)
Another spawn today, 12 days since the last spawn. I opened the gate yesterday, but she was in no mood at all to go over onto his side. He went over to her side, and repeatedly tried to lead her over to his side, but no go. After about 10 minutes, he gave up and went back home. Today, they spent the morning together at the divider, and when I opened the gate, she almost immediately went over to his side. Within about 1 minute, she was in his capped PVC cave, and again, she stayed in there for about 20 minutes or so. He was either in there with her, or guarding the entrance, the whole time. Almost as soon as she exited the spawning cave, she returned to her side of the divider.
I have only one larvae left from the 2/21 hatch at this point. I'll keep maintaining that BRT as long as that larvae survives. I'll prepare another BRT for today's spawn.
|
|
|
 Re:Breeding Journal, Species: Pseudochromis tapeinosoma
Thursday, March 1, 2012 1:21 PM
Re:Breeding Journal, Species: Pseudochromis tapeinosoma
Thursday, March 1, 2012 1:21 PM
( permalink)
With regards to die-off: Maybe pH change from the dark period and the live algae? O2 change from the same?
--Andy, the bucket man. "Not to know the mandolin is to argue oneself unknown...." --Clara Lanza, 1886
|
|
|
 Re:Breeding Journal, Species: Pseudochromis tapeinosoma
Saturday, March 3, 2012 11:02 PM
Re:Breeding Journal, Species: Pseudochromis tapeinosoma
Saturday, March 3, 2012 11:02 PM
( permalink)
OK, it's official. I'm an idiot. I am officially too stupid to live. I got mixed up between the hatch days of these dottybacks and the pipefish. I've spent the last two days again doing numerous small water changes on the broodstock tank to make the water in the broodstock tank as much like my new, aged ASW as possible. Tonight, I got the BRT all set up, put about 5 gallons of the new, aged ASW into it, made sure temp and SG matched the broodstock tank, got the tumbler set up, rotifers and copepods ready, right after lights out, I pulled the eggs, and placed them in the tumbler. I took a tiny sample to get some microscope pics of the eggs right before hatch. Hmm. These don't look quite ready. Still large yolk sacks present. Hmm. No eggs hatching. Hmm. I check this journal, to discover the spawn happened on 29 February -- only three days ago! Doh! I quickly replaced the eggs into Dad's PVC cave. The good news is, he is such a good Dad! He's continuing to care for the eggs, and hasn't eaten them on me! At least I got pictures of the eggs at 3 1/2 days (84 Hours Post Spawn). Here they are: 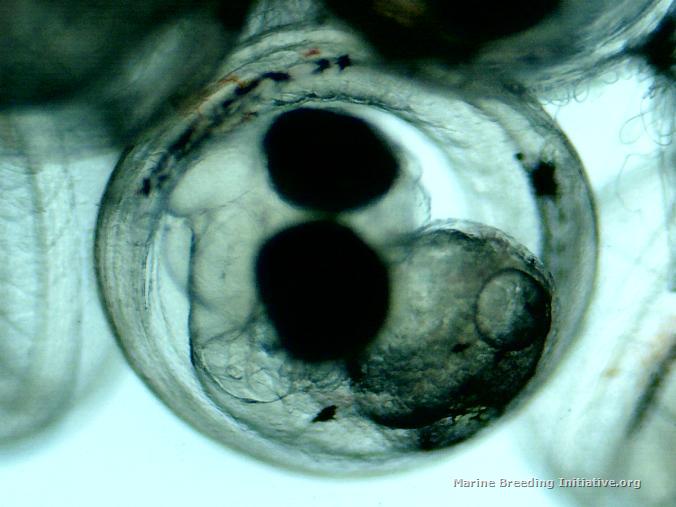 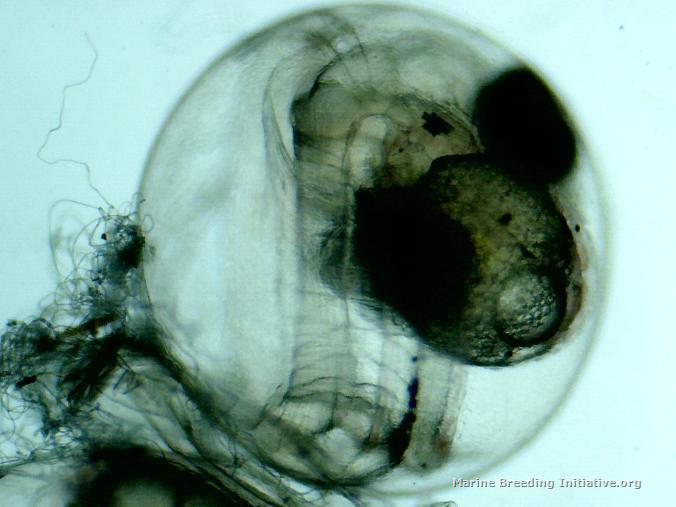 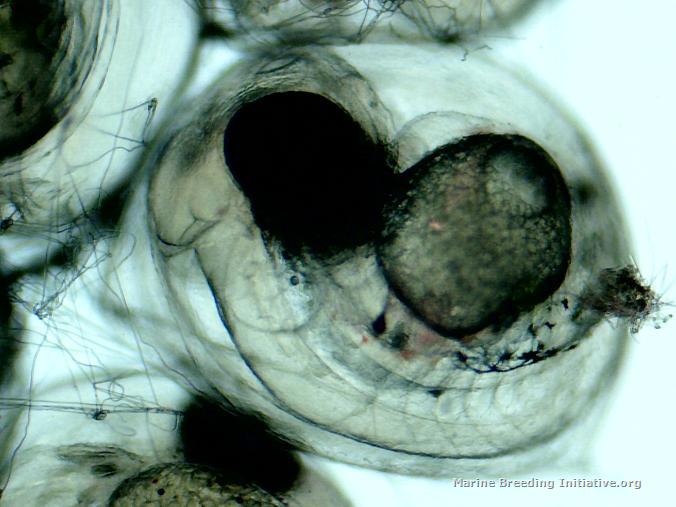 Now, I have to go set up the snagger for the stupid pipes!
|
|
|
 Re:Breeding Journal, Species: Pseudochromis tapeinosoma
Sunday, March 4, 2012 7:11 AM
Re:Breeding Journal, Species: Pseudochromis tapeinosoma
Sunday, March 4, 2012 7:11 AM
( permalink)
Those are some lovely pictures, Jim. I hope the boy carries through with egg tending.
--Andy, the bucket man. "Not to know the mandolin is to argue oneself unknown...." --Clara Lanza, 1886
|
|
|
 Re:Breeding Journal, Species: Pseudochromis tapeinosoma
Sunday, March 4, 2012 10:58 AM
Re:Breeding Journal, Species: Pseudochromis tapeinosoma
Sunday, March 4, 2012 10:58 AM
( permalink)
Thanks, Andy! As of this morning, Dad is still doing his job. He does look at me with a more jaundiced eye, though!
|
|
|
 Re:Breeding Journal, Species: Pseudochromis tapeinosoma
Sunday, March 4, 2012 11:01 AM
Re:Breeding Journal, Species: Pseudochromis tapeinosoma
Sunday, March 4, 2012 11:01 AM
( permalink)
If his eyes are yellow then I have to ask, what have you been putting in the water?  You know that peeing in the tank as an ammonia source is a bad idea, right? 
--Andy, the bucket man. "Not to know the mandolin is to argue oneself unknown...." --Clara Lanza, 1886
|
|
|
 Re:Breeding Journal, Species: Pseudochromis tapeinosoma
Sunday, March 4, 2012 11:23 PM
Re:Breeding Journal, Species: Pseudochromis tapeinosoma
Sunday, March 4, 2012 11:23 PM
( permalink)
|
|
|
 Re:Breeding Journal, Species: Pseudochromis tapeinosoma
Monday, March 5, 2012 7:03 AM
Re:Breeding Journal, Species: Pseudochromis tapeinosoma
Monday, March 5, 2012 7:03 AM
( permalink)
I love how the fish use the gate, very cute! Great pics too Jim. Good luck with the new batch. 
|
|
|
|