Change Page: 12 > | Showing page 1 of 2, messages 1 to 20 of 31 - powered by ASPPlayground.NET Forum Trial Version
Reports tied to this Journal
Author
|
Message

|
 Culture Journal, Species: [Dunaliella tertiolecta]
Tuesday, January 28, 2014 9:52 PM
Culture Journal, Species: [Dunaliella tertiolecta]
Tuesday, January 28, 2014 9:52 PM
( permalink)
Culturing Journal DataSheet
This first post should be updated regularly to include new information as events take place or changes are made to your system General Species: Dunaliella tertiolecta Species description: "Cells permanently green, radially symmetrical, ellipsoidal, oval or rarely ovate or pyriform. Cells 5-18 µm in length (mean 9.4�12.4 �m), 4.5�14.0 �m wide (mean 7.1-8.2 µm)." - http://www.algaebase.org/search/species/detail/?species_id=51616 Culture source (link if possible ): http://florida-aqua-farms../shop/microalgae-disks/ If algae, CCMP # (Optional ): http://ccmp.bigelow.edu/ Culture Establishment Date: Continuation Date: Culturing Vessel Details Salinity: 1.028 Temperature: Ambient, around 75-78F pH: Not Measured Vessel description: 500ml canning jar Lighting description: 3 Lights of America Linkable Fluorescent Undercabinet Portable Lights Lighting cycle: 22 hours on, 2 hours off Aeration description: Turbulent Methodologies Split methodology: Every day, 25-50% split per culture Culture medium description:
1. Add 2x Guillards F/2 formula per 100ml of culture water
2. Heat in microwave until 212F has been achieved
3. Allow cooling until room temperature
4. Split algae into containers
5. Run airline into culture with a mild-turbulent flow
6. Have warm lights on the culture running at 22 hours on, 2 hours off
7. Split every day or two, or until the jar has a light green tint to it
8. Repeat 2-6 for every split Cell count: (if known) Reference links: Additional Information (No Pictures or Videos in the Section Please) Notes: You will be required to provide photographic evidence and as much detail as possible about your project in this thread.
If your thread does not contain detailed enough photos and information the MBI Council will not be able to approve your reports.
<message edited by Amphispur on Tuesday, March 11, 2014 9:57 PM>
|
|
|
 Re:Culture Journal, Species: [Dunaliella tertiolecta]
Tuesday, January 28, 2014 10:10 PM
Re:Culture Journal, Species: [Dunaliella tertiolecta]
Tuesday, January 28, 2014 10:10 PM
( permalink)
I am going to try and culture this, partially just to see how hard/how it is done, and also to try and feed to my copepods too. The only true "culturing" information I could find on it was a paper entitled: Culture of the Marine Phytoplankter, Dunaliella tertiolecta, with Light-Dark Cycles. Within this paper, it gave me a "basic" way to culture it. Essentially what it had said was that A.) A "Guillard 'F' culture medium was employed throughout." I'm fairly certain that can just be called our Guillard F2 formula, but please note me if those are two different things. B.) Autoclaving everything that holds or touches algae. I do not have the tools to autoclave my materials, so I will be using a water sterilizing method and using freshly cleaned containers throughout the culture. C.) The dilution of the culture as done at the end of each "Dark Period" (aka a few minutes or so before the lights came on) to around 50,000 cells/ml. I will try my best to get a moderately accurate density, I have a FAF Microalgae Density Stick to measure it with. D.) The "best" way to culture it was on a 24 hour light cycle. I'm not sure how I feel about this, I may just try different cultures, one with the full 24 hours, and one with around 20 hours or so of light. E.) Their lights were around 550-600 m µ, which I'm not sure what that means so if anyone cares to tell me what m µ is I would be very grateful! I will be ordering the culture in the next couple of days, and working on from there. I think I will try some different containers with these. I will do one batch of 24 hour with a "round" mason jar (Later on I will post the exact size of it) and a more cylindrical container and see how those results vary day by day. As far as split cycle goes, it is starting to sound like a split should be done every day, which will get tedious at times, but I will get through it.
<message edited by Amphispur on Tuesday, January 28, 2014 11:24 PM>
|
|
|
 Re:Culture Journal, Species: [Dunaliella tertiolecta]
Tuesday, January 28, 2014 11:14 PM
Re:Culture Journal, Species: [Dunaliella tertiolecta]
Tuesday, January 28, 2014 11:14 PM
( permalink)
Nice to see you working on something unusual and interesting, Zach!
A) Guillard F/2 is simply Guillard F at 1/2 strength. For F strength, just use twice as much F/2, and voila, you have F.
B) Just do what you have already been doing with other phytoplankton cultures, and be as clean and sterile, and cautious about contamination as possible.
C) Get a hemocytometer, and learn how to use it. Meanwhile, your Secchi stick will do just fine.
D) Knock yourself out.
E) The 550-600 millimicrons is an indication of the main wavelength in the light used (although the abstract of the paper I read said 580-590 millimicrons). That wavelength corresponds to a very yellow-orange color, indicating a pretty low color temperature. I recommend that you use 6500K lights, although that wavelength would actually tend to suggest something even more warm white (lower Kelvin) than the 6500K.
Good luck with them!
|
|
|
 Re:Culture Journal, Species: [Dunaliella tertiolecta]
Tuesday, January 28, 2014 11:23 PM
Re:Culture Journal, Species: [Dunaliella tertiolecta]
Tuesday, January 28, 2014 11:23 PM
( permalink)
Thanks Jim! Ah ok, that makes sense on the F/2. I will get a hemocytometer as soon as I can. I'll run by home depot in the next couple of days and see if I can find any bulbs that would best fit that description.
Thanks again Jim!
|
|
|
 Re:Culture Journal, Species: [Dunaliella tertiolecta]
Thursday, February 6, 2014 5:52 PM
Re:Culture Journal, Species: [Dunaliella tertiolecta]
Thursday, February 6, 2014 5:52 PM
( permalink)
Awesome to see you doing this. Have you started this up yet? I wonder how this culture actually starts from the disk. Will it be like the nanno disk that starts fine, or will it be like the Iso disk where it is actually nearly impossible for people to get a culture going off of it? I am actually waiting to see how it works for you before I order some replacements for the ones my dog ate...
|
|
|
 Re:Culture Journal, Species: [Dunaliella tertiolecta]
Thursday, February 6, 2014 6:01 PM
Re:Culture Journal, Species: [Dunaliella tertiolecta]
Thursday, February 6, 2014 6:01 PM
( permalink)
Hey shannpeach! I believe that it will be arriving tomorrow, since they only ship on Thursdays or something like that. One question for you guys, that I probably should have asked from the beginning. So when doing a culture from a disk (I've only done their starter culture in a liquid form, probably should have requested that but too late now) how do you guys start them up? Do you just stick the container in the bottom of your first culture container then after it has grown, take it out?
|
|
|
 Re:Culture Journal, Species: [Dunaliella tertiolecta]
Thursday, February 6, 2014 6:06 PM
Re:Culture Journal, Species: [Dunaliella tertiolecta]
Thursday, February 6, 2014 6:06 PM
( permalink)
They will include really detailed instructions with the starter. From what I remember, you put a bit of water on the disk and leave it with the lid on for a day or something, then use a sterile Q tip thing (which they usually send) to get the algae off the plate. Then put it in your culture flask.
Or something along those lines. It's pretty easy...when it works lol
|
|
|
 Re:Culture Journal, Species: [Dunaliella tertiolecta]
Thursday, February 6, 2014 6:08 PM
Re:Culture Journal, Species: [Dunaliella tertiolecta]
Thursday, February 6, 2014 6:08 PM
( permalink)
Sounds easy enough lol, if it turns out to crash, I'll get another culture and this time put in a request for a liquid form of the algae, I believe that they do that for most all algaes.
|
|
|
 Re:Culture Journal, Species: [Dunaliella tertiolecta]
Thursday, February 6, 2014 8:35 PM
Re:Culture Journal, Species: [Dunaliella tertiolecta]
Thursday, February 6, 2014 8:35 PM
( permalink)
So shannpeach, apparently my dad (the man whom had actually ordered the algae for me) DID actually order it to come in a liquid form from them, so should be smooth sailing 
|
|
|
 Re:Culture Journal, Species: [Dunaliella tertiolecta]
Friday, February 7, 2014 8:31 AM
Re:Culture Journal, Species: [Dunaliella tertiolecta]
Friday, February 7, 2014 8:31 AM
( permalink)
Well that's good news for you, but bad news for me! hahaha So much for you being my guinea pig... I'll still be following along to see how this goes 
|
|
|
 Re:Culture Journal, Species: [Dunaliella tertiolecta]
Friday, February 7, 2014 8:54 AM
Re:Culture Journal, Species: [Dunaliella tertiolecta]
Friday, February 7, 2014 8:54 AM
( permalink)
Shannpeach, what were you going to try and culture it for? I'm mainly doing this to see how my tisbes do on it as a food source, not sure how well it would be benificial for us culturing it since around 30% or so of its body mass is oil.
|
|
|
 Re:Culture Journal, Species: [Dunaliella tertiolecta]
Friday, February 7, 2014 2:05 PM
Re:Culture Journal, Species: [Dunaliella tertiolecta]
Friday, February 7, 2014 2:05 PM
( permalink)
There are some invert related breeding papers(urchins, sea hares, etc) that used this species so I thought it may be a good culture to have handy for those sorts of projects if I decide to work on them.
|
|
|
 Re:Culture Journal, Species: [Dunaliella tertiolecta]
Friday, February 7, 2014 2:11 PM
Re:Culture Journal, Species: [Dunaliella tertiolecta]
Friday, February 7, 2014 2:11 PM
( permalink)
Ah! I may have to try this now too  That is if my ceriths decide if they ever want to spawn again lol.
|
|
|
 Re:Culture Journal, Species: [Dunaliella tertiolecta]
Thursday, February 20, 2014 12:27 PM
Re:Culture Journal, Species: [Dunaliella tertiolecta]
Thursday, February 20, 2014 12:27 PM
( permalink)
Well.... Still has not arrived yet  Going to contact them today and see what's going on...
|
|
|
 Re:Culture Journal, Species: [Dunaliella tertiolecta]
Saturday, March 8, 2014 3:51 PM
Re:Culture Journal, Species: [Dunaliella tertiolecta]
Saturday, March 8, 2014 3:51 PM
( permalink)
Well, time for some exciting news! Culture JUST arrived in the mail today, think they just lost our last order or something. First off, HOLY COW THESE GUYS ARE TINY! I got a hemocytometer, as you recommended Jim, and these guys are not that motile, so they are fairly easy to count. First pic is of the culture it came in from FAF. Few pics afterwards is of it under my Celestron 44340, at 100x (for some reason my celestron doesn't read 400x that well, anyone else have that problem?) I'll try and see if I can convince my science teacher to let me use his dissection microscope to see if it helps. Mother Culture from FAF:  At 40x: 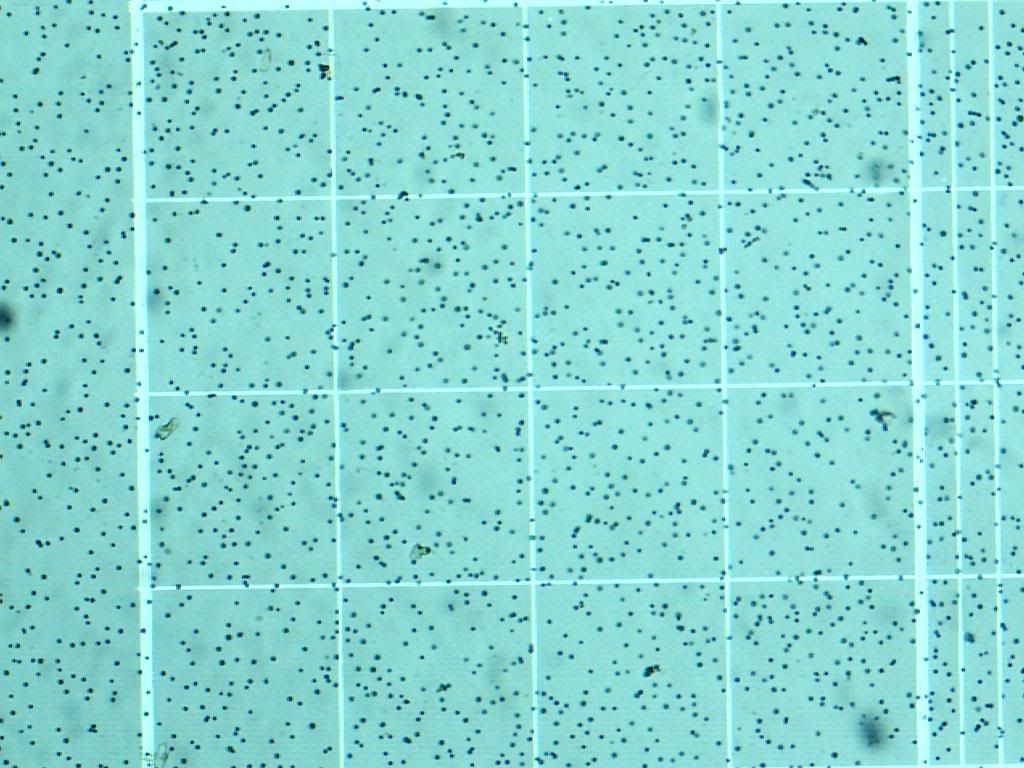 At 100x: 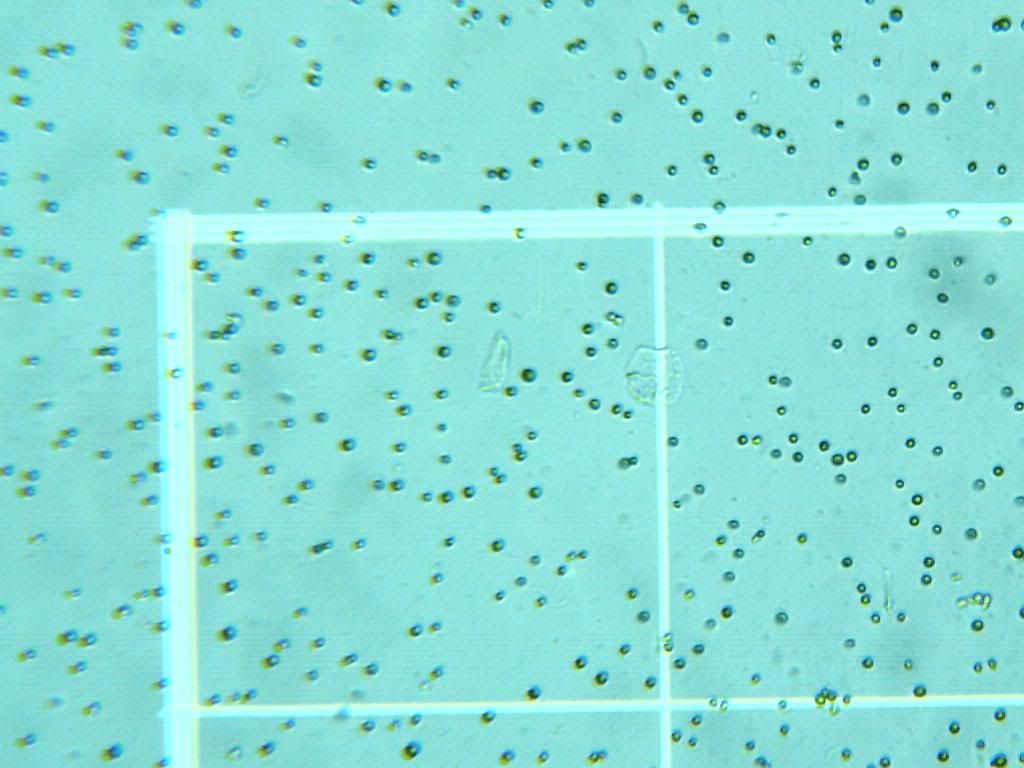 Also, as I am updating this now, I'm still working on setting up the algae and will put up more of what the first "trail" of this species is.
<message edited by Amphispur on Saturday, March 8, 2014 4:30 PM>
|
|
|
 Re:Culture Journal, Species: [Dunaliella tertiolecta]
Saturday, March 8, 2014 4:06 PM
Re:Culture Journal, Species: [Dunaliella tertiolecta]
Saturday, March 8, 2014 4:06 PM
( permalink)
Also, I know the cells don't look right, which has me a tad worried. this species is suppost to be "tear-drop" shaped instead of circular, so I'll play around with it under the microscope and see if I can get that 400x to atleast give me cell shape
|
|
|
 Re:Culture Journal, Species: [Dunaliella tertiolecta]
Saturday, March 8, 2014 4:43 PM
Re:Culture Journal, Species: [Dunaliella tertiolecta]
Saturday, March 8, 2014 4:43 PM
( permalink)
So, onto experiment #1: Seeing as to how these guys explode overnight, (seen in Culture of the Marine Phytoplankter, Dunaliella tertiolecta, with Light-Dark Cycles) I took a chance and split the culture into three mason jars, all of which have fresh water I boiled earlier (I treated the water with Jim's microwave-kill-everything-method, see my Isochrysis journal for more detail) for 45 minutes, since it was only three jars. Now, the experiment itself. I labeled each jar 1, 2, and 3. Jar 1 has a mild aeration, just enough to get a small current through, ~10-15 bubbles per second. Jar 2 has a VERY rapid aeration, more of a turbulent water. Jar 3 has a very calm aeration, bubble per second. I'm going to leave them be for today after this, since the explode overnight I'll check them in the morning and see how they are doing. 
|
|
|
 Re:Culture Journal, Species: [Dunaliella tertiolecta]
Sunday, March 9, 2014 10:46 AM
Re:Culture Journal, Species: [Dunaliella tertiolecta]
Sunday, March 9, 2014 10:46 AM
( permalink)
Well, didn't get the "explosion" I was hoping to get. They are still alive, just not very dense. This could have been because they only had around 5-6 hours of light, then they went out. I'll be putting them on a separate timer today set at 22hrs on, 2 hours off. Hopefully that should escalate the growth more. I didn't see that much if a difference in cell numbers between 1, 2, or 3, but again, there wasn't much growth in the first place.
|
|
|
 Re:Culture Journal, Species: [Dunaliella tertiolecta]
Monday, March 10, 2014 6:03 PM
Re:Culture Journal, Species: [Dunaliella tertiolecta]
Monday, March 10, 2014 6:03 PM
( permalink)
Well, today went MUCH better with the culture. I set the lights so that they went off around noonish, and back on at 2pm, so that there were 22 hrs on, and I got back home in time to see how it resulted. And there was a TON of growth! As you can see in the photos, Jar 2 had the best growth. Because of this, I'll be using turbulent water throughout the culture. Next experiment, how many days until split? Essentially, one jar split every day, one jar every two, one every three, and one every four days. These splits won't happen til tomorrow morning, due to the need to microwave the jars (which I totally forgot to do today!) So far though, I've managed not to crash it, GOOD SIGN!   Also, I know I already mentioned earlier about how they should be split every day, but I notice that this culture is not as dark as when it arrived to me, so I'm curious
|
|
|
 Re:Culture Journal, Species: [Dunaliella tertiolecta]
Monday, March 10, 2014 6:55 PM
Re:Culture Journal, Species: [Dunaliella tertiolecta]
Monday, March 10, 2014 6:55 PM
( permalink)
Well, played around with the hemocytometer to calculate cell density, and got for chamber 1: 330 cells, chamber 2: 260 cells, chamber 3: 267 cells, and chamber 4 with 307. I averaged that amount, (average was 291) and did that to the 10^4, which was 2.91 x10^6 cells/ml, or 2,910,000 cells/ml. Which means a ROUGH estimate of how many cells in the container would be around 8,261,665,000 cells in the entire container, each mason jar was filled to about 12 cups (measurements on the side,) convert that to ml, 2,839.06, then multiplied 2,839.06 * 2,910,000. Jim, can you double-check my calculations on that? I'm fairly certain I did them correctly, but I just want to be sure. Also, although those may seem like big numbers, it is no where near as dense as when I first got it, proving the point of how these guys need more time.
|
|
|
|