Change Page: < 1234567 > | Showing page 2 of 7, messages 21 to 40 of 130 - powered by ASPPlayground.NET Forum Trial Version
Author
|
Message

|
 Re:Breeding Journal, Species: Pseudochromis tapeinosoma
Thursday, January 26, 2012 11:29 AM
Re:Breeding Journal, Species: Pseudochromis tapeinosoma
Thursday, January 26, 2012 11:29 AM
( permalink)
Thanks for the suggestions, Matt. For now, I'm keeping what I have set up. What are your concerns about air? FWIW, as you can see in the side view of my tumbler, I have a short length of vinyl tubing with some net mesh stretched over the top end and a rubber band securing the mest shoved into the neck of the funnel so as to make a screen to prevent the egg ball from falling into the neck (which was a problem before I did this). The screen tends to cause the air to accumulate beneath it, and then it gets released in small "burps" about once per second. The egg ball has been tumbling nicely for about 24 hours now. I haven't removed any eggs to check with the microscope, but both last night, and this morning, I removed the egg ball with a spoon, and examined it briefly with a high-power magnifying glass. As of this morning, I can definitely see embryos developing! They are looking right on schedule, as far as the image Mindy found and posted here: http://www.mbisite.org/Fo.ms/fb.ashx?m=64320 I'd say that last night, they were all looking like F or G, but this morning, they are more like K or L! EDIT: Looks like someone at Advanced Aquarist has, within the last 24 hours, noticed Mindy's link to their image, and is blocking access to it. To see the image I mean, you now have to visit their site. Here's the url: http://www.advancedaquari...8/10/breeder#section-8
<message edited by JimWelsh on Friday, January 27, 2012 2:31 PM>
|
|
|
 Re:Breeding Journal, Species: Pseudochromis tapeinosoma
Thursday, January 26, 2012 1:32 PM
Re:Breeding Journal, Species: Pseudochromis tapeinosoma
Thursday, January 26, 2012 1:32 PM
( permalink)
OK, so I broke down, and took a small sample to check with the microscope. Definite embryo development: 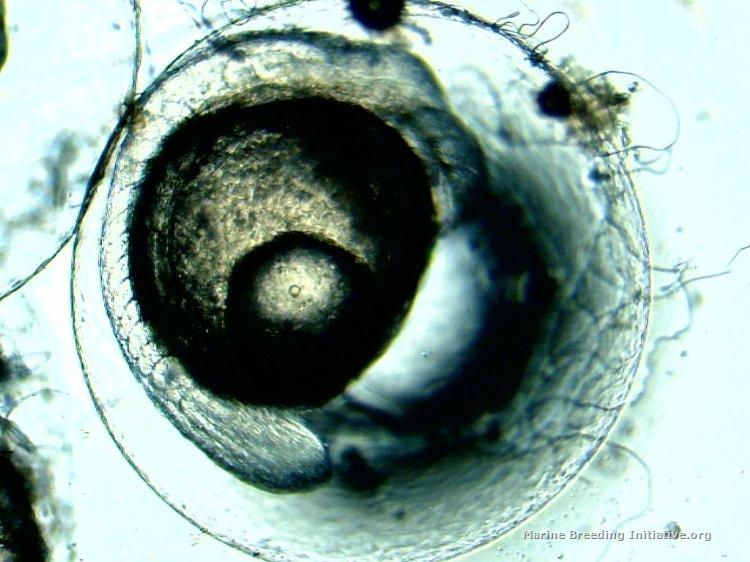
|
|
|
 Re:Breeding Journal, Species: Pseudochromis tapeinosoma
Thursday, January 26, 2012 2:58 PM
Re:Breeding Journal, Species: Pseudochromis tapeinosoma
Thursday, January 26, 2012 2:58 PM
( permalink)
OK, now that I've sacrificed a few eggs to the microscope gods, the problem with using air to tumble the ball that Tal warned me about on MBI Chat last night is happening -- the egg ball is getting big air bubbles trapped inside it, making it bob on the surface. SO, I've dusted off my Witt-style tumbler that I made way back when and documented on another breeding site, and now have them, well, not exactly tumbling, but more like levitating in the water:  The eggs aren't really "tumbling", but a constant stream of water is flowing past the egg ball, and it is gently swaying to and fro. I hope that is sufficient.
|
|
|
 Re:Breeding Journal, Species: Pseudochromis tapeinosoma
Thursday, January 26, 2012 3:07 PM
Re:Breeding Journal, Species: Pseudochromis tapeinosoma
Thursday, January 26, 2012 3:07 PM
( permalink)
Should be good Jim - I've noticed that as long as the "edges" are getting flow that will carry over - if you can actually get full rotation, that's even better.
|
|
|
 Re:Breeding Journal, Species: Pseudochromis tapeinosoma
Thursday, January 26, 2012 5:32 PM
Re:Breeding Journal, Species: Pseudochromis tapeinosoma
Thursday, January 26, 2012 5:32 PM
( permalink)
Awesome progress Jim! It is so cool to almost hear your excitement! Could you describe the water tumbler please?
|
|
|
 Re:Breeding Journal, Species: Pseudochromis tapeinosoma
Thursday, January 26, 2012 5:44 PM
Re:Breeding Journal, Species: Pseudochromis tapeinosoma
Thursday, January 26, 2012 5:44 PM
( permalink)
Google the phrase "my version of witt's tumbler". Should come up as the first link.
|
|
|
 Re:Breeding Journal, Species: Pseudochromis tapeinosoma
Thursday, January 26, 2012 6:30 PM
Re:Breeding Journal, Species: Pseudochromis tapeinosoma
Thursday, January 26, 2012 6:30 PM
( permalink)
Got it, thanks! It looks very much like the first air tumbler I ever made. You mention it is described in Witt's book, I didn't notice before so I will have to take a closer look. 
|
|
|
 Re:Breeding Journal, Species: Pseudochromis tapeinosoma
Friday, January 27, 2012 1:22 PM
Re:Breeding Journal, Species: Pseudochromis tapeinosoma
Friday, January 27, 2012 1:22 PM
( permalink)
The water tumbler is consistently doing a very good job of keeping the egg ball suspended and awash with freshly oxygenated water. This morning, examining the eggs with a high-power magnifying glass, I can see definite embryo development, right on schedule. I can clearly see tails and rows of dots made up by the developing vertebrae. I even saw some movement in a few of them! There are some white eggs, too, but right now, the developing eggs appear to outnumber the white eggs by about a 2:1 ratio, at least. I don't want to disturb the egg ball, and so am not going to sample any for microscope photos, but I did try to get some macro shots with the bellows. I had a very hard time with lighting and focus, but here is the best shot I could get today in the limited time I had (blue arrows point at a couple of tails / spines that can be seen): 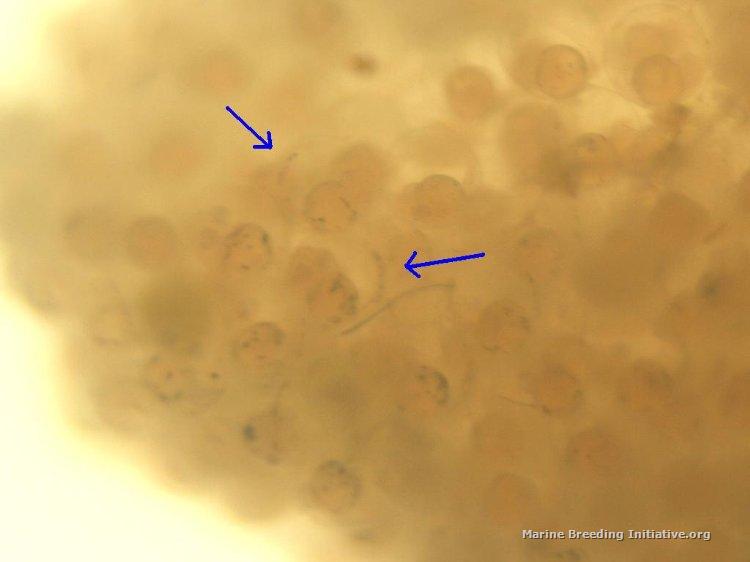
|
|
|
 Re:Breeding Journal, Species: Pseudochromis tapeinosoma
Friday, January 27, 2012 2:13 PM
Re:Breeding Journal, Species: Pseudochromis tapeinosoma
Friday, January 27, 2012 2:13 PM
( permalink)
now that's pretty cool. Nice share!
RLTW 180 Gallon Mixed Reef Then I heard the voice of the Lord saying, "Whom shall I send? And who will go for us?" And I said, "Here am I. Send me!" Isaiah 6:8
|
|
|
 Re:Breeding Journal, Species: Pseudochromis tapeinosoma
Friday, January 27, 2012 2:45 PM
Re:Breeding Journal, Species: Pseudochromis tapeinosoma
Friday, January 27, 2012 2:45 PM
( permalink)
This. Is. Exciting!!!!!!!!! Keep up the good work!!!!
Jonathan Foster
FishEye Aquaculture
|
|
|
 Re:Breeding Journal, Species: Pseudochromis tapeinosoma
Friday, January 27, 2012 3:05 PM
Re:Breeding Journal, Species: Pseudochromis tapeinosoma
Friday, January 27, 2012 3:05 PM
( permalink)
(evil cackles coming from this side of the scream).
|
|
|
 Re:Breeding Journal, Species: Pseudochromis tapeinosoma
Friday, January 27, 2012 9:41 PM
Re:Breeding Journal, Species: Pseudochromis tapeinosoma
Friday, January 27, 2012 9:41 PM
( permalink)
After getting home from work tonight, I see that the white eggs are winning. The still alive and viable eggs are exclusively on the bottom of the egg ball. I'm pretty darn sure that there is simply not enough flow in my tumbler, and that I need to either increase the flow and/or re-engineer the tumbler altogether. I've added a bit of Hydrogen Peroxide per the instructions on a Facebook chat session with an experienced breeder. Those instructions were as follows:
Take 1 part 3% Hydrogen peroxide, and add that to 9 parts RO/DI water. Mix well. Add 1 ml of that to each 5 gallons of water in the system where the tumbler is. NOTE: this assumes the tumbler is in a separate, isolated system, and not part of a larger system, or in a display. This dosage will give appx. 0.15 PPM of H2O2. Dose as necessary, but do not exceed 0.5 PPM H2O2.
I've dosed 2 ml in the 5 gal system that the tumbler is in, for appx. 0.3 PPM H2O2. We'll see if that helps.
As of tonight, there still are several eggs with clearly viable embryos inside them (I can see them moving from time to time), but it is only about 1/4 of the total egg number now, and perhaps even less than that.
We'll see if I get any of these to hatch. If not, then maybe Dad will be able to do his job next time, and maybe "next time" will come soon....
|
|
|
 Re:Breeding Journal, Species: Pseudochromis tapeinosoma
Friday, January 27, 2012 9:53 PM
Re:Breeding Journal, Species: Pseudochromis tapeinosoma
Friday, January 27, 2012 9:53 PM
( permalink)
|
|
|
 Re:Breeding Journal, Species: Pseudochromis tapeinosoma
Saturday, January 28, 2012 6:20 AM
Re:Breeding Journal, Species: Pseudochromis tapeinosoma
Saturday, January 28, 2012 6:20 AM
( permalink)
Jim for what it's worth, I've had much better success with dealing with fungus when I stopped using Hydrogen peroxide, and switched to Formalin. More specifically, I've been using Kordon's Rid Ich Plus. It contains Formalin @ 11.25% and malachite green. Dosing is simple - 10 drops per gallon. It seemed that there were always different results from Hydrogen Peroxide be it for clowns, dotties, wrasses, basslets, etc. Very, very rarely do I see fungus taking over eggs now, and those that are white don't spread with any kind of speed. Hope it helps! (Oh and a 16oz bottle has lasted me nearly four months, and is still half full 
|
|
|
 Re:Breeding Journal, Species: Pseudochromis tapeinosoma
Saturday, January 28, 2012 8:22 AM
Re:Breeding Journal, Species: Pseudochromis tapeinosoma
Saturday, January 28, 2012 8:22 AM
( permalink)
Novice question: Aren't you supposed to remove the white eggs?
|
|
|
 Re:Breeding Journal, Species: Pseudochromis tapeinosoma
Saturday, January 28, 2012 8:26 AM
Re:Breeding Journal, Species: Pseudochromis tapeinosoma
Saturday, January 28, 2012 8:26 AM
( permalink)
You can for clowns and pelagic spawners - dotties and other fish that have an egg mass it is next to impossible to get the white ones out that are on the inside, unless you'd like to chop the mass up and surgically remove the fungus eggs. And it takes a long time too 
|
|
|
 Re:Breeding Journal, Species: Pseudochromis tapeinosoma
Saturday, January 28, 2012 11:44 AM
Re:Breeding Journal, Species: Pseudochromis tapeinosoma
Saturday, January 28, 2012 11:44 AM
( permalink)
Yeah, the dottyback eggs are all tied together with this very strong thread that reminds me of spider web, it is so strong.
It's probably too late for this batch. The whole ball is looking rather fuzzy white today. There are a few viable eggs still, but I doubt they're going to make it until tomorrow night! I'll add Malachite Green to the shopping list (I seem to remember reading somewhere that you can't get it in California, because of the Formalin being a carcinogen, but I may be mistaken).
The broodstock continues to do well, and she is plumping up again nicely. I let her out to play today, and the male was doing tons of leading towards his cave, and showed very little aggression. The female did just want to get back home, though -- she clearly isn't ready yet. It is curious -- just like Matt has observed, once they make it through the PVC gate, they can't seem to find their way back home, even though they are circling the entrance that they just came through. I always have to net them to get them back onto their own side, although sometimes the female will hide from the net in the male's PVC cave, and stay in there while I just move the cave over to her side, and then she swims out.
|
|
|
 Re:Breeding Journal, Species: Pseudochromis tapeinosoma
Saturday, January 28, 2012 2:05 PM
Re:Breeding Journal, Species: Pseudochromis tapeinosoma
Saturday, January 28, 2012 2:05 PM
( permalink)
try methylene blue, it's what I used to use for angle fish eggs (fresh water). It will aid in oxygen transport across the egg membrane as well. I don't think MG does that. If you are looking for it mixed with formalin, use that ick stop (I think that's what it's called), comes in a small blue-green plastic bottle. Too bad you don't live by me, I have a pound of MG in my basement.
|
|
|
 Re:Breeding Journal, Species: Pseudochromis tapeinosoma
Saturday, January 28, 2012 2:25 PM
Re:Breeding Journal, Species: Pseudochromis tapeinosoma
Saturday, January 28, 2012 2:25 PM
( permalink)
What dosage of MB? What it says on the bottle? Reason I ask is my wife got some MB from the LFS yesterday, and this morning I dosed at the recommended rate for preventing fungus in fish eggs (10 drops / gallon). Turned the water DARK BLUE. The movement in the eggs stopped. Before the MB, I could see movement in some of the eggs almost continuously, and if I watched one egg, it would predictably move within a minute or so. After the MB, no more movement.  After observing that, I did three 95% water changes with seasoned SW at the same SG and temp as the egg tank water. Still, no more movement is being seen.    EDIT: Bear in mind, this batch was probably a lost cause anyway, but for the future, a safe and effective treatment protocol using Methylene Blue would be helpful, if anybody has one.
<message edited by JimWelsh on Saturday, January 28, 2012 2:47 PM>
|
|
|
 Re:Breeding Journal, Species: Pseudochromis tapeinosoma
Saturday, January 28, 2012 3:29 PM
Re:Breeding Journal, Species: Pseudochromis tapeinosoma
Saturday, January 28, 2012 3:29 PM
( permalink)
What is recommended is 1 to 3 ppm in the tank. It will kill biofilter bacteria. This should make a light to medium blue water, but you still should be able to see the eggs. Don't know why your fish should be so sensitive to it, but it is usually added at the time the eggs are layed and concentration slowly decreases with time as the embryos develop.
|
|
|
|